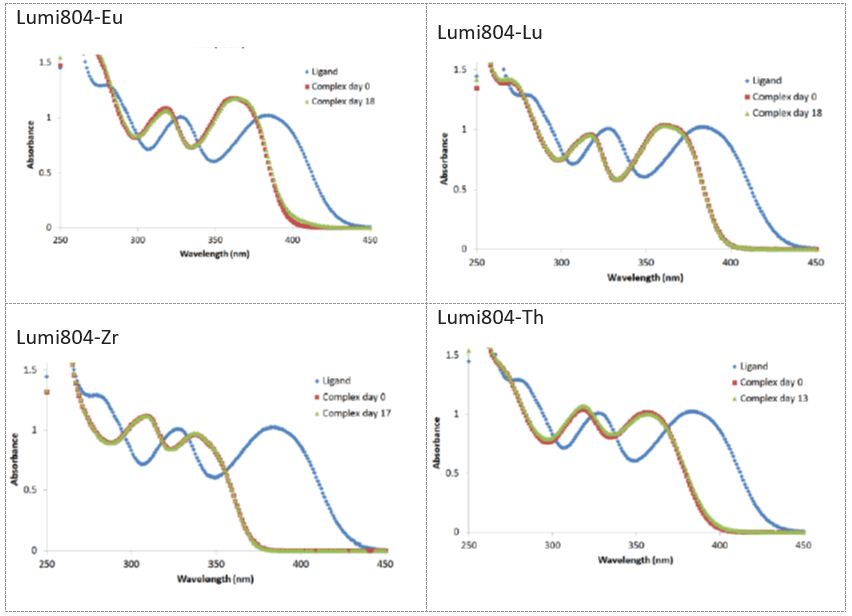
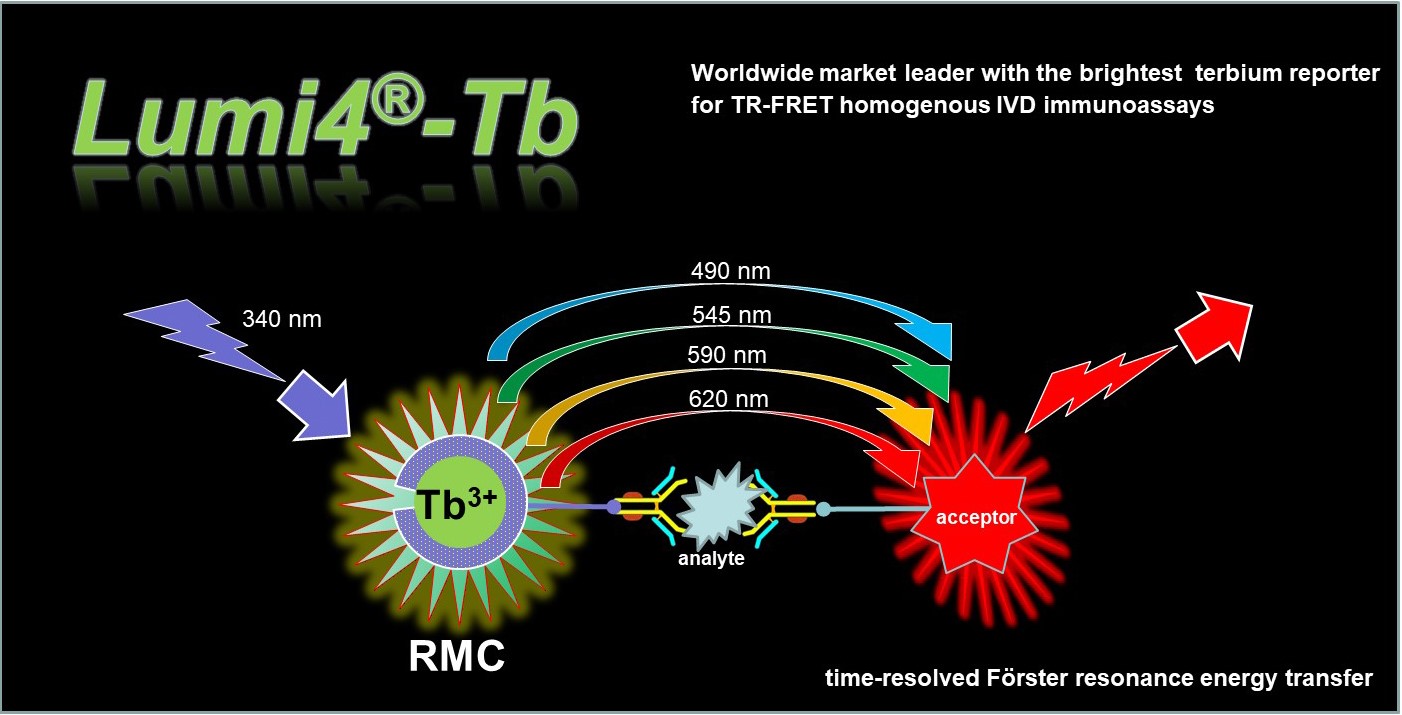
Lumiphore’s chelation technology meets the unmet need of rapid, stable chelation at or below physiological temperature with exceptional in-vivo stability for a wide range of radionuclides. Our mBFC technology is the next generation of chelation technology with fundamental features surpassing DOTA, DTPA, and DFO for radiotheranostic applications.
Our proprietary chelation platform, paired with an appropriate targeting vector (such as an antibody, antibody fragment, protein or peptide), facilitates reliable localization of radionuclides to biological sites of interest.
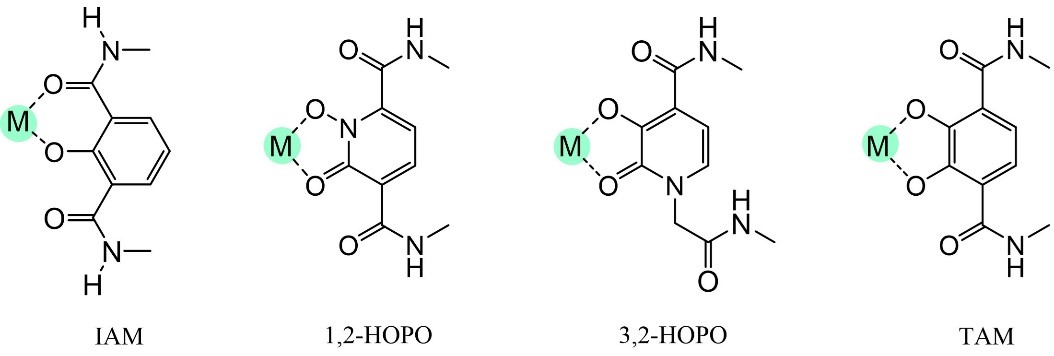
The company’s proprietary ‘Raymond Chelation Technology’ reworks Nature’s siderophore components into macrocyclic Bi-Functional Chelators (mBFCs), suitable for:
These macrocyclic BFCs are best-in-class materials for reliable incorporation and delivery of metal ion payloads in medicine.